39 in the sketch of the structure of nf3 label all bonds.
EOF SO3 Molecular Geometry, Lewis Structure, and Polarity Explained When we draw it, firstly we get the three structures at the top. Sulfur in the center and Oxygen around it is making a connection (each) to the central atom. There should be single bonds initially. There are 6 electrons in the structure which we have made, and there are 24 in total. So we need to put 18 electrons down.
Dipoles & Dipole Moments: Molecule Polarity - Study.com Here are some quick steps to determine the dipole moment of the molecule: Step 1) Obtain the Lewis dot structure for the molecule. Step 2) Draw dipoles for each bond. Dipole arrows point towards...
In the sketch of the structure of nf3 label all bonds.
PCl3 Molecular Electron Geometry, Lewis Structure, Bond Angles and ... Phosphorus trichloride is made up of one Phosphorus atom and three Chlorine atoms, having a chemical formula of PCl3. It is a volatile liquid that reacts with water and releases HCl gas. It is a toxic compound but is used in several industries. Phosphorus Trichloride is widely used in manufacturing Phosphites and other organophosphorus compounds. Geometrical Structure of Molecules and Ions using Molecular ... Show 3-D sketch of the structure and label all bond angles. H. O. H. 109.5o. How many sigma bonds? How many pi bonds? 2 σ and 0 π bonds. Is the substance an.11 pages SF6 Molecular Geometry, Lewis Structure, Shape, and Polarity In Lewis Structure, we show the bonds in the structure by drawing a straight line between two atoms. So all these bonds will take up 12 valence electrons out of 48. Place all the remaining valence electrons around the Fluorine atoms and check if the octets of all the fluorine atoms are complete.
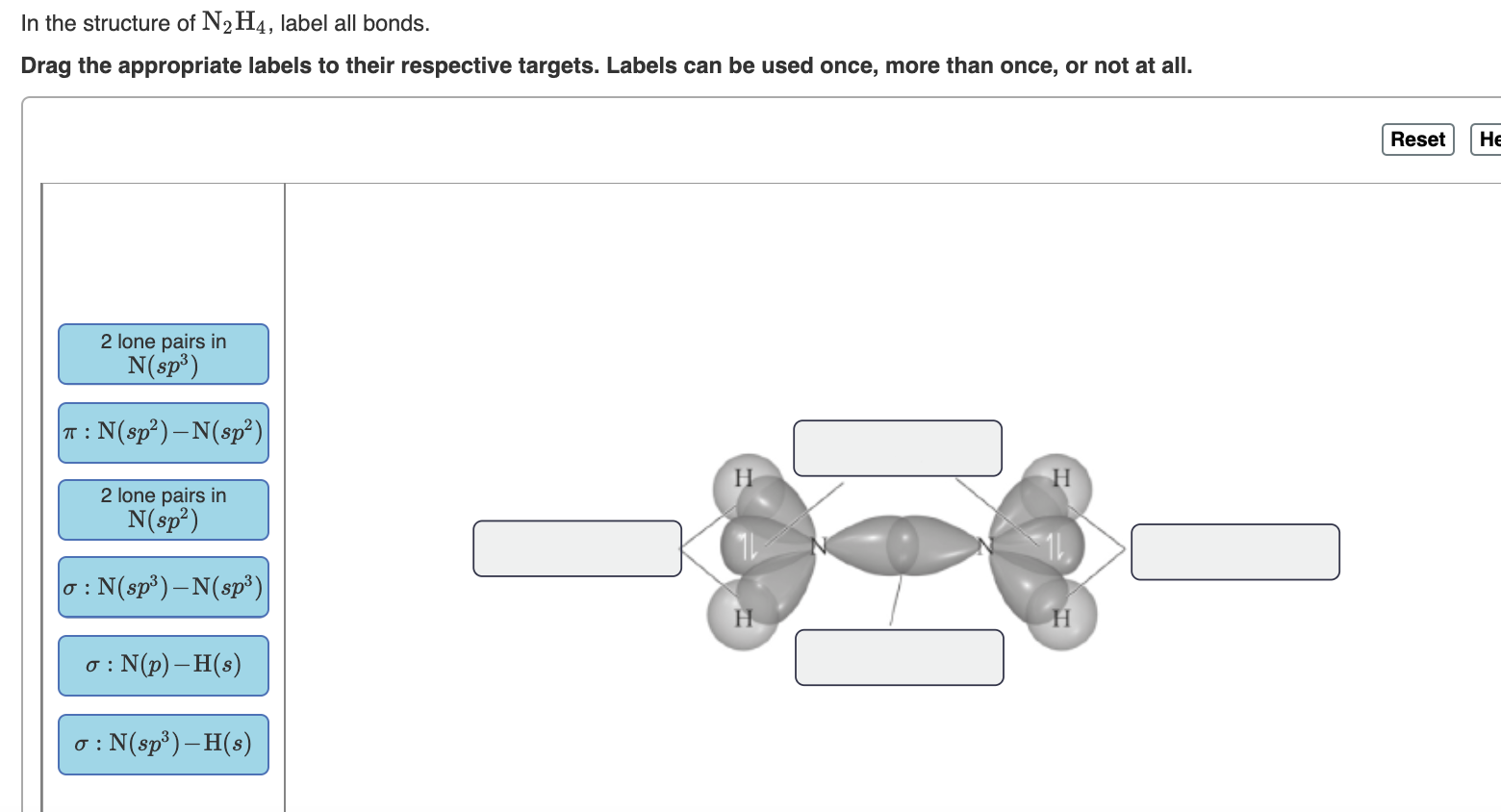
In the sketch of the structure of nf3 label all bonds.. Molecule Bonding Hybridization For Scheme A Write Each And To accommodate the two lone pairs and the bonding pair, it will also form three equivalent sp^2 hybrid orbitals C2h2 Hybridization Diagram Write a hybridization and bonding scheme for each of the following molecules In valence bond theory, each bond results from the overlap of two atomic orbitals on adjacent • Define the term hybridization and name the types of hybrid orbitals used by atoms ... Lewis Structures: Learn How to Draw Lewis Structures | Albert.io Determine the total number of valence electrons to be depicted in the Lewis diagram. Example: CO 2 Total = 16 Step 2. Place least electronegative element in center and draw single bonds from the central atom to other atoms. Step 3. Determine how many electrons must be added to central element. NH3 Lewis Structure, Molecular Geometry, Hybridization, Bond Angle & Shape NH3 Bond angles There are three single bonds and one lone pair of electrons in NH3 molecule. It has a molecular geometry of trigonal pyramidal which also looks like a distorted tetrahedral structure. The shape is distorted because of the lone pairs of electrons. This pair exerts repulsive forces on the bonding pairs of electrons. HCN Lewis Structure, Molecular Geometry, Shape, and Polarity Once we know the Lewis structure and Molecular Geometry of any molecule, it is easy to determine its bond angles and polarity. As this molecule has a linear molecular geometry, HCN has bond angles of 180 degrees. HCN Shape As both Hydrogen and Nitrogen are placed far from each other at bond angles of 180 degrees, it forms a linear shape.
CHE 105/110 - Introduction to Chemistry - Textbook The concept that atoms play a fundamental role in chemistry is formalized by the modern atomic theory, first stated by John Dalton, an English scientist, in 1808. It consists of three parts: All matter is composed of atoms. Atoms of the same element are the same; atoms of different elements are different. Answered: In the sketch of the structure of NF3… | bartleby In the sketch of the structure of NF3 label all bonds. Drag the appropriate labels to their respective targets. Labels can be used once, more than once, ...1 answer · Top answer: Step 1 structure of NF3 is: Nitrogen has five valance electrons in its valence shell. It requires 3 more electrons to complete its octet. So, it forms ... CH3Cl Lewis Structure, Molecular Geometry, Bond ... - Geometry of Molecules It has 14 valence electrons, and all of them participate in forming bonds. In the Lewis structure of CH3Cl, Carbon is at the central position and all the other atoms around it. The bond angles of Carbon with Hydrogen and Chlorine atoms are 109.5 degrees. This molecule has a tetrahedral shape, and the central carbon atom has sp3 hybridization. In the Lewis structure for NF3, the total number of lone pairs ... The molecular formula for nitromethane is CH3NO2 a. Draw a valid Lewis structure for this molecule. Label one of the oxygens on your structure (a) and the other (b). valince bonding e a8 non-bonding e: O / 0 l o l b. Indicate the hybridization of the... Posted 7 months ago Plagiarism Checker
Write a hybridization and bonding scheme for each molecule ... Sketch the molecule, including overlapping orbitals, and label all bonds using the notation shown in Examples 6.1 and 6.2. a. CH2Br2 b. SO2 c. NF3 d. BF3 ...4 answers · Top answer: So for this first part part A. We need to form four bonds to carbon. And since we have four ... SF6 Molecular Geometry, Lewis Structure, Shape, and Polarity In Lewis Structure, we show the bonds in the structure by drawing a straight line between two atoms. So all these bonds will take up 12 valence electrons out of 48. Place all the remaining valence electrons around the Fluorine atoms and check if the octets of all the fluorine atoms are complete. Geometrical Structure of Molecules and Ions using Molecular ... Show 3-D sketch of the structure and label all bond angles. H. O. H. 109.5o. How many sigma bonds? How many pi bonds? 2 σ and 0 π bonds. Is the substance an.11 pages PCl3 Molecular Electron Geometry, Lewis Structure, Bond Angles and ... Phosphorus trichloride is made up of one Phosphorus atom and three Chlorine atoms, having a chemical formula of PCl3. It is a volatile liquid that reacts with water and releases HCl gas. It is a toxic compound but is used in several industries. Phosphorus Trichloride is widely used in manufacturing Phosphites and other organophosphorus compounds.


Post a Comment for "39 in the sketch of the structure of nf3 label all bonds."