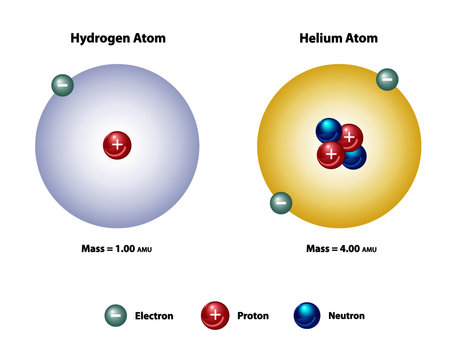
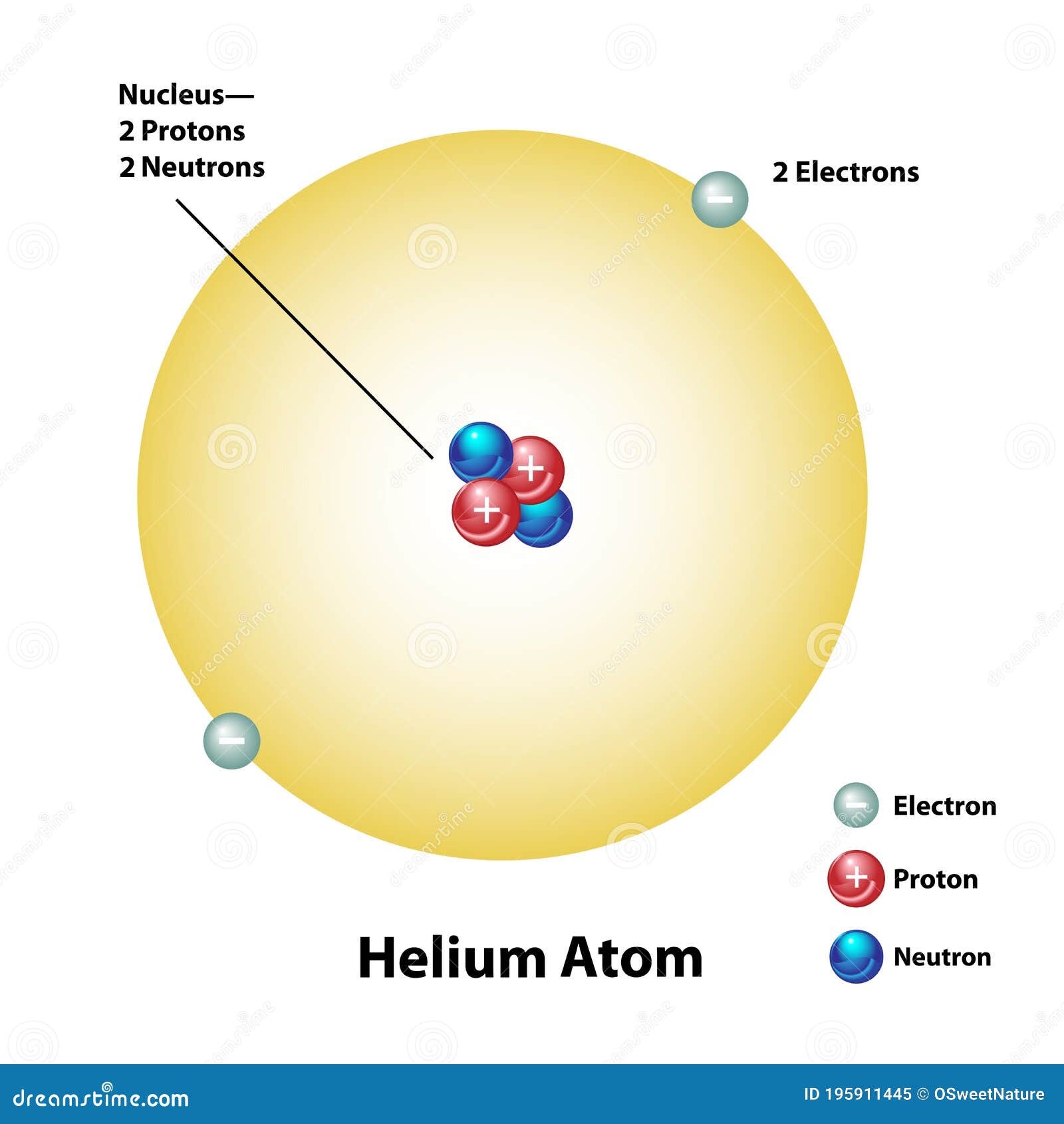
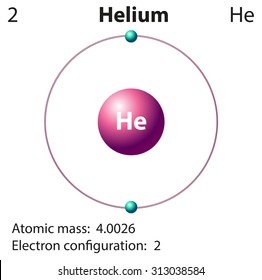
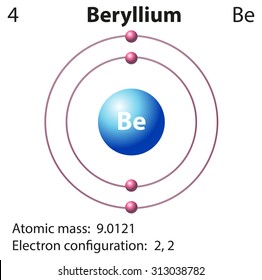
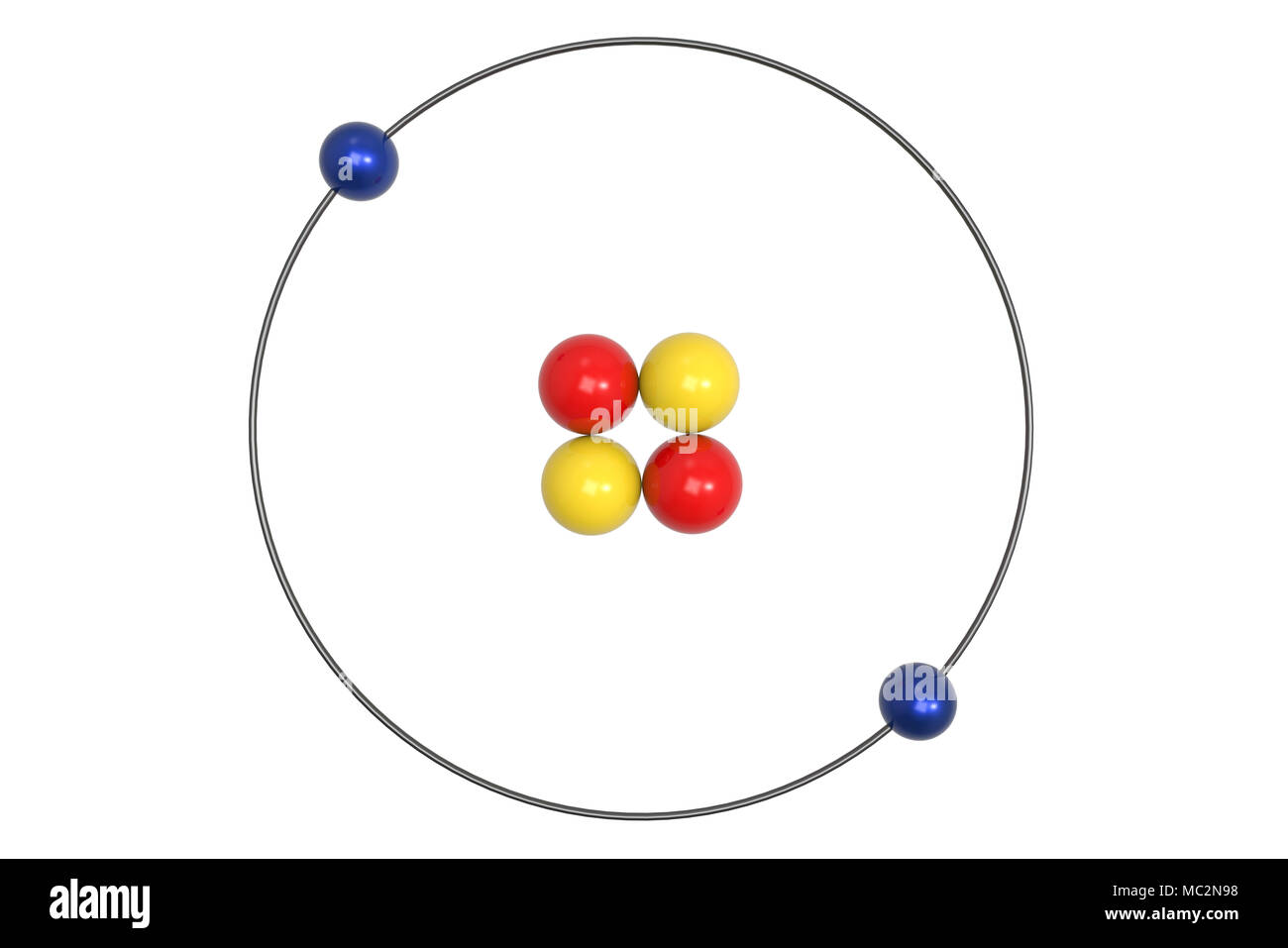
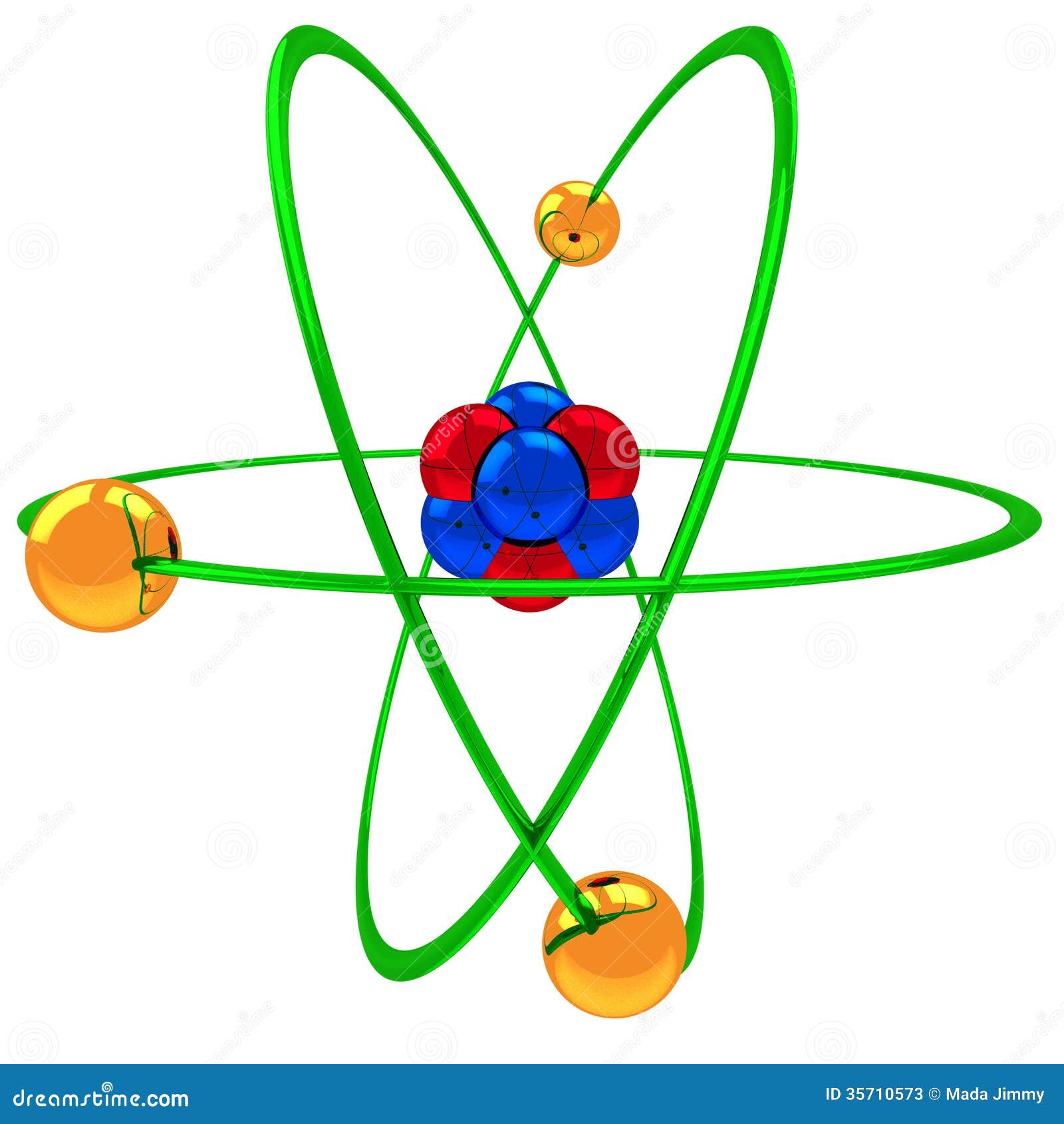
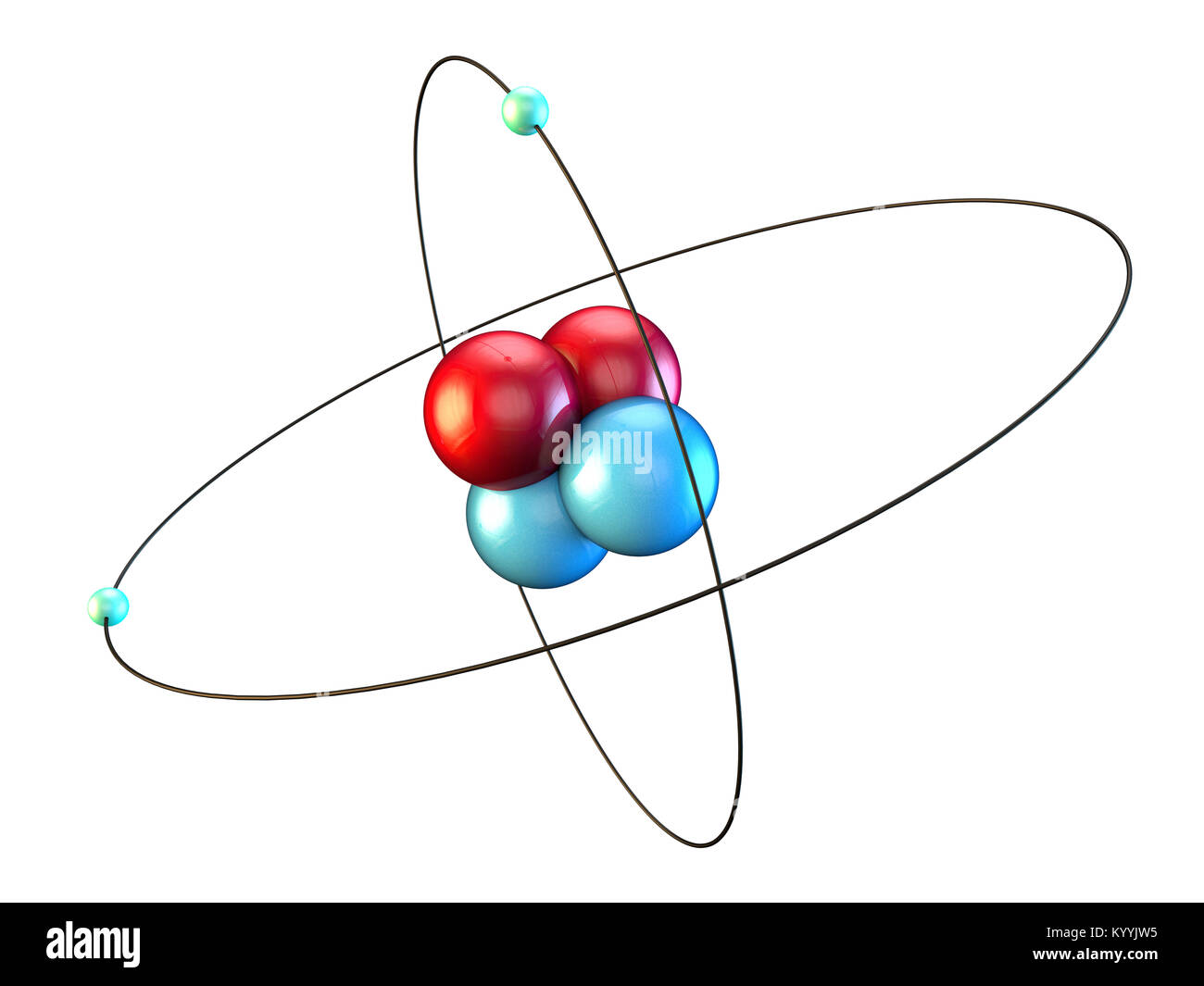
43 helium atom diagram labeled
en.wikipedia.org › wiki › Molecular_orbital_diagramMolecular orbital diagram - Wikipedia For a diatomic molecule, an MO diagram effectively shows the energetics of the bond between the two atoms, whose AO unbonded energies are shown on the sides. For simple polyatomic molecules with a "central atom" such as methane (CH 4) or carbon dioxide (CO 2), a MO diagram may show one of the identical bonds to the central atom. For other ... Table of Elements and Chemistry In 1913, chemistry and physics were topsy-turvy. Some big hitters - including Mendeleev - were talking seriously about elements lighter than hydrogen and elements between hydrogen and helium. Visualizing the atom was a free-for-all, and Mendeleev's justification for a periodic table based on atomic weights was falling apart at the seams.
What are Stereoisomers? - Study.com 26.02.2022 · Learn about stereoisomer definition, types, and examples. Understand the difference between constitutional isomers and stereoisomers, and...
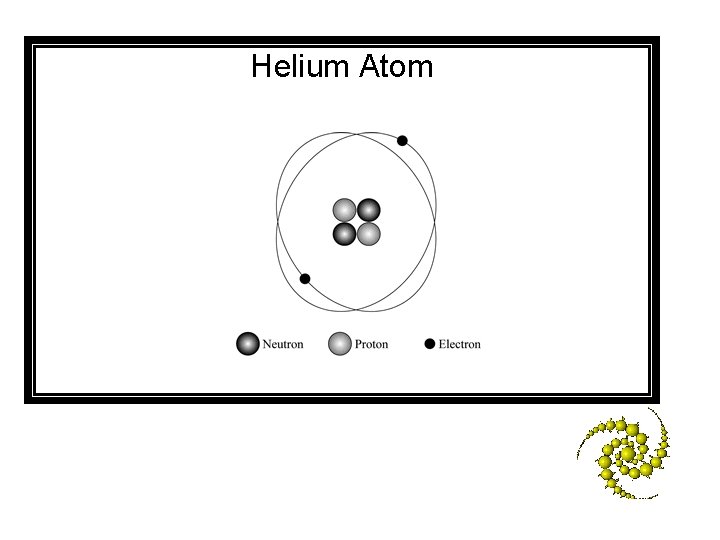
Helium atom diagram labeled
Build an Atom - Atoms | Atomic Structure | Isotope Symbols - PhET Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Then play a game to test your ideas! › class-11-chemistry-chapter-2Structure of Atoms Class 11 Notes Chemistry Chapter 2 Apr 30, 2021 · Bohr’s Model For Hydrogen Atom Postulates: 1. The electron in the hydrogen atom can move around the. the nucleus in a circular path of fixed radius and energy. These paths are called orbits, stationary states, or allowed energy states. 2. The energy of an electron in the orbit does not change with time. phet.colorado.edu › en › simulationBuild an Atom - Atoms | Atomic Structure | Isotope Symbols ... Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Then play a game to test your ideas!
Helium atom diagram labeled. Structure of Atoms Class 11 Notes Chemistry Chapter 2 - Learn … 30.04.2021 · Structure of Atoms Notes Class 11 Chemistry Chapter 2. Sub-Atomic Particles: Dalton’s Atomic Theory regarded the atom as the ultimate particle of matter. It explained satisfactorily various laws of chemical combination like the law of conservation of mass, the law of constant composition, and the law of multiple proportions. Periodic table with Charges Labeled on it (7 HD Images) 09.07.2022 · Ionic charge: When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). This electric charge generated on the ion is known as Ionic charge. When atoms gain electron/s, the negatively charged ion is formed, and when the atoms lose electron/s, the positively charged ion is formed. periodictableguide.com › periodic-table-with-chargesPeriodic table with Charges Labeled on it (7 HD Images) Jul 09, 2022 · Periodic table Labeled with Everything (9+ different Images) Periodic table with Ionization Energy values (labeled image) Periodic table with Electronegativity values (labeled image) Periodic table with Valence Electrons Labeled (7 HD Images) Periodic table with Charges Labeled on it (7 HD Images) Molecular orbital diagram - Wikipedia A molecular orbital diagram, or MO diagram, ... Carbon is the central atom of the molecule and a principal axis, the z-axis, is visualized as a single axis that goes through the center of carbon and the two oxygens atoms. For convention, blue atomic orbital lobes are positive phases, red atomic orbitals are negative phases, with respect to the wave function from the solution of the ...
CHAPTER4 Carbon and its Compounds - National Council of … 42. Catenation is the ability of an atom to form bonds with other atoms of the same element. It is exhibited by both carbon and silicon. Compare the ability of catenation of the two elements. Give r easons. 43. Unsaturated hydr ocarbons contain multiple bonds between the two C-atoms and show addition reactions. Give the test to Bohr model - Wikipedia The Bohr model gives almost exact results only for a system where two charged points orbit each other at speeds much less than that of light. This not only involves one-electron systems such as the hydrogen atom, singly ionized helium, and doubly ionized lithium, but it includes positronium and Rydberg states of any atom where one electron is far away from everything else. en.wikipedia.org › wiki › Bohr_modelBohr model - Wikipedia The Bohr model gives almost exact results only for a system where two charged points orbit each other at speeds much less than that of light. This not only involves one-electron systems such as the hydrogen atom, singly ionized helium, and doubly ionized lithium, but it includes positronium and Rydberg states of any atom where one electron is far away from everything else. Periodic Table of Elements and Chemistry In 1913, chemistry and physics were topsy-turvy. Some big hitters - including Mendeleev - were talking seriously about elements lighter than hydrogen and elements between hydrogen and helium. Visualizing the atom was a free-for-all, and Mendeleev's justification for a periodic table based on atomic weights was falling apart at the seams.
periodictableguide.com › periodic-table-withPeriodic table with Valence Electrons Labeled (7 HD Images) Jul 09, 2022 · There are many more labeled Periodic Table of valence electrons given below. But before that, just have a look at the concept of valence electrons. Valence electrons: The electrons present in the outermost orbit of an atom are called valence electrons. Periodic table with Valence Electrons Labeled (7 HD Images) 09.07.2022 · The above image clearly shows you the Periodic table with Valence Electrons labeled on it. You can also get the printable Periodic table with valence electrons, from this article only. Skip to content. Periodic Table Guide Menu. Popular contents; About; Periodic table with Valence Electrons Labeled (7 HD Images) July 9, 2022 by Admin. The above image … X-Ray Photoelectron Spectroscopy - an overview - ScienceDirect R.R. Mather, in Surface Modification of Textiles, 2009 X-ray photoelectron spectroscopy. X-ray photoelectron spectroscopy (XPS) is a quantitative technique for measuring the elemental composition of the surface of a material, and it also determines the binding states of the elements. XPS normally probes to a depth of 10 nm. However, because XPS is an ultra-high vacuum … phet.colorado.edu › en › simulationBuild an Atom - Atoms | Atomic Structure | Isotope Symbols ... Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Then play a game to test your ideas!
› class-11-chemistry-chapter-2Structure of Atoms Class 11 Notes Chemistry Chapter 2 Apr 30, 2021 · Bohr’s Model For Hydrogen Atom Postulates: 1. The electron in the hydrogen atom can move around the. the nucleus in a circular path of fixed radius and energy. These paths are called orbits, stationary states, or allowed energy states. 2. The energy of an electron in the orbit does not change with time.
Build an Atom - Atoms | Atomic Structure | Isotope Symbols - PhET Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Then play a game to test your ideas!




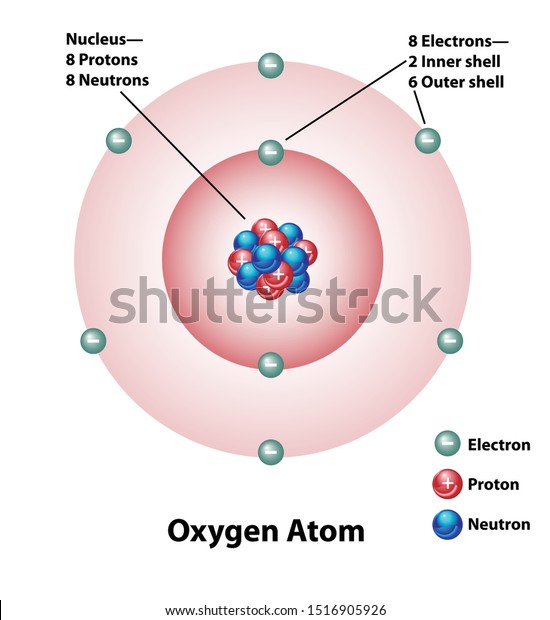

![0.2 The atom, Chemistry grade 10 [caps], By OpenStax (Page 10 ...](https://www.jobilize.com/ocw/mirror/col11303_1.4_complete/m38126/CG10C3_005.png)
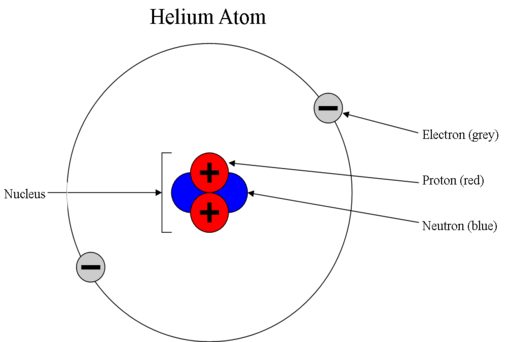
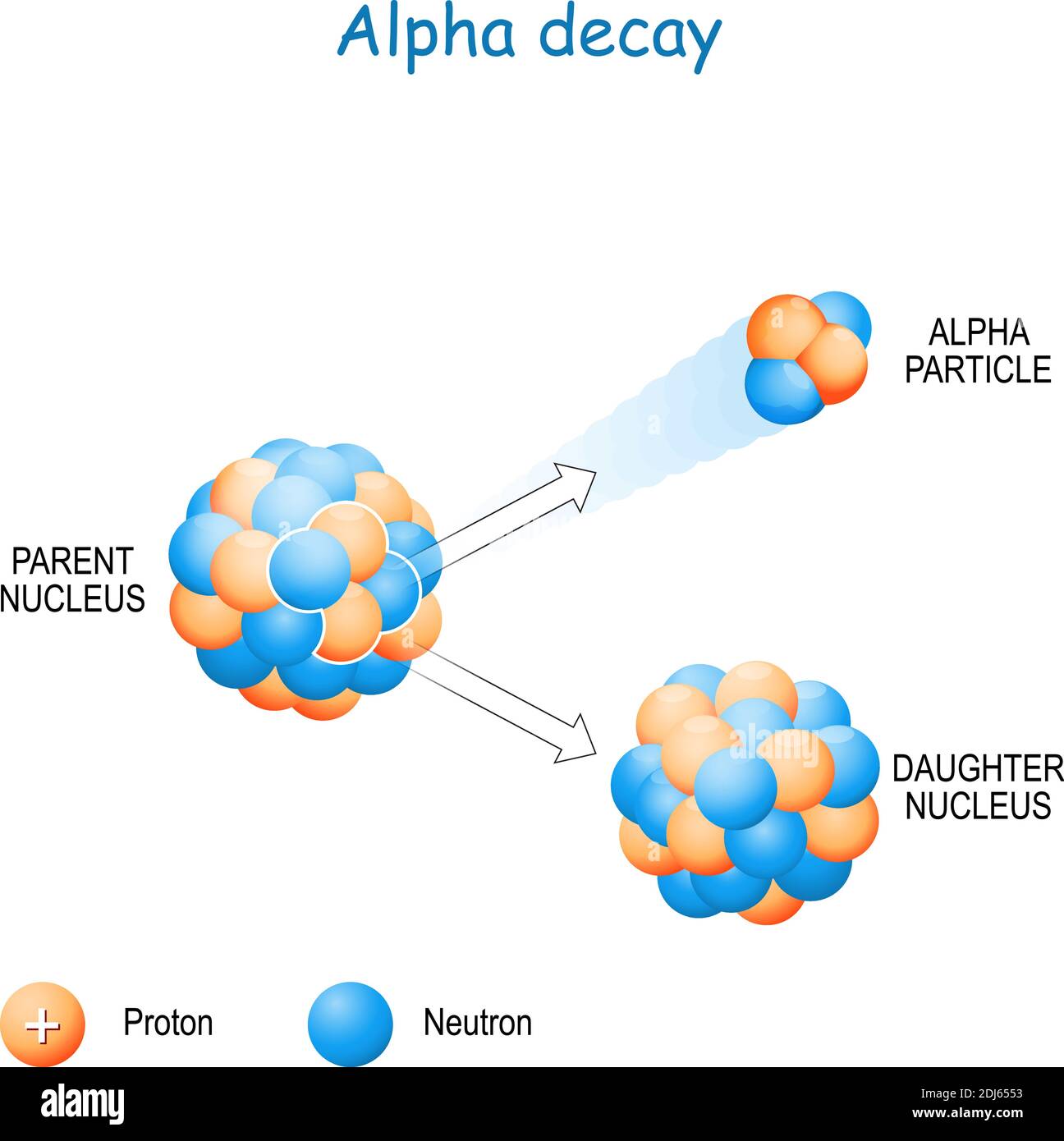






















Post a Comment for "43 helium atom diagram labeled"